-
口蹄疫 —— 防控详细指南 Foot and Mouth Disease - Details Regarding Prevention & Control
2026-05-02
本文将深入探讨口蹄疫的一些方面,这些方面此前尚未得到充分讨论。
In this article we discuss details of FMD which have not been discussed in as much depth -
2022-12-12
In this article we discuss details of FMD which have not been discussed in as much depth
口蹄疫 —— 防控详细指南
Foot and Mouth Disease - Details Regarding Prevention & Control
作者:吉施德,(美国职业兽医博士,公共卫生硕士); 高级顾问 - 美牧生
Author: Stephan Gibson, DVM MPH; Senior Consultant – Mei Mu Sheng
过去几周,已有许多文章报道了口蹄疫的防控措施。这些文章大多集中于动物隔离、限制人员和车辆进入牧场及消毒等重要措施。
Over the past few weeks, many articles have been written regarding prevention and control measures of foot-and-mouth disease (FMD). These have generally focused on quarantine of animals, restriction of people and vehicles entering farms, and disinfection, which are very important.
我们假设本文读者已对口蹄疫的基本概念、防控方面有所了解,因此不再赘述相关基础知识。本文将深入探讨口蹄疫的一些其它方面,这些方面此前尚未得到充分讨论。我们希望本文的某些内容能够对进一步优化口蹄疫的防控工作有所助益。
We assume that those reading this article already have a basic understanding of what FMD is and how to prevent and control it. Therefore, we will not spend time reviewing the basics. Instead, in this article we discuss other details of FMD, aspects which have not been discussed in as much depth. We hope that some of these may prove useful in further improving the prevention and control of the disease.
首先,我想澄清一些常见的误解:
口蹄疫不会传染给人类。对人类健康没有危害,因此与动物接触的人无需测量体温或担心自身健康。但是人上呼吸道可能携带病毒,并通过呼吸将其传播给易感动物(Sellers et al., 1971; Sellers & Forman, 1973)。这种风险虽然不高,但并非不可能。
南非1型(SAT-1) 与非洲猪瘟 (ASF) 完全不同,它只是口蹄疫病毒的一个血清型,最初在南非被发现。该病毒传播方式和临床症状与口蹄疫相似,并不是与非洲猪瘟相似。虽然两种病毒的名称中都包含“非洲”(且都能感染猪),但它们本质完全不同。
First of all, I want to address some misunderstandings that I have seen:
Foot-and-mouth disease is not contagious to people. There is no risk to human health, so there is no reason for people in contact with animals to check their temperature or otherwise be concerned for their own health. However, people can harbor the virus in their upper respiratory tract and transmit it to susceptible animals through their breath (Sellers et al., 1971; Sellers & Forman, 1973). This is not a very high risk, but it is possible.
SAT-1 is nothing like African Swine Fever (ASF). It is simply a serotype of FMD which was first identified in South Africa. The modes of transmission and clinical disease are like FMD, not like ASF. While both viruses have "Africa" in the name (and both can infect pigs), they are completely different.
需要注意的是,血清型和毒株之间存在差异,而且大多数参考的研究都是针对口蹄疫A型、O型、亚洲1型(Asia-1)和C型的。因此,虽然SAT-1型可能与之类似,但我们目前还没有太多关于SAT-1型的确凿研究。
Note that there are differences between serotypes and strains, and that most of the research referenced has been done on FMD types A, O, Asia1, and C. Therefore, while SAT-1 will likely be similar, we do not have much definitive research on SAT-1.
传播途径 Transmission
最开始感染途径 Initial Infection Route
牛羊通常通过吸入密切接触的其他动物的感染性飞沫或气溶胶而感染(Brown et al., 2022),而猪很难通过飞沫或气溶胶感染。猪通常通过采食感染性物质而感染,并且需要更大的病毒载量才能成功感染(Donaldson et al., 2001)。这种差异可能是由于病毒在牛和水牛体内的主要复制部位是鼻咽,而在猪体内的主要复制部位是口咽(Arzt et al., 2024; Stenfeldt et al., 2020)。病毒通常需要通过吸入才能感染鼻咽。
人们对绵羊和山羊的感染途径了解较少,但似乎与牛最为相似。
Cattle and sheep are usually infected by inhalation of infectious droplets or aerosols from other animals in close contact (Brown et al., 2022), while it is very difficult for pigs to be infected by droplets or aerosols. Pigs are usually infected by ingestion of infectious materials, and require a much larger dose of virus to be successfully infected (Donaldson et al., 2001). This difference is probably due to the fact that the primary site of virus replication in cattle and buffalo is the nasopharynx, and in pigs it is the oropharynx (Arzt et al., 2024; Stenfeldt et al., 2020). In order for virus to infect the nasopharynx, it generally needs to be inhaled.
Less is known about the infection route for sheep and goats, but it appears to be most similar to cattle.
病毒排出 Shedding
已知猪通过呼吸排出的病毒量远高于牛羊,因此它们是口蹄疫传播给牛羊的高风险群体(Donaldson et al., 1970, 2001)。
已知牛会通过牛奶和粪便排出大量病毒(Sellers & Forman, 1973)。
已知绵羊和山羊即使临床症状轻微,也会排出病毒(Donaldson et al., 1970)。
Pigs are known to shed much larger amounts of virus than cattle and sheep in their breath, making them high risks for transmitting FMD to cattle and sheep (Donaldson et al., 1970, 2001).
Cattle are known to shed large amounts of virus in milk and feces (Sellers & Forman, 1973).
Sheep and goats are known to shed virus even though they have minimal clinical signs (Donaldson et al., 1970).
传播途径 Transmission Routes
与受感染动物直接接触(包括飞沫传播)是主要的传播途径(Brown et al., 2022)。通过污染物传播也是一种风险。通过控制人员和动物进出牧场,以及对带入牧场的车辆、衣物、工具等进行消毒,可以非常有效地控制这些风险。
Direct contact with an infected animal (including droplet transmission) is the primary mode of transmission (Brown et al., 2022). Transmission by fomites is also a risk. By controlling the movement of animals and people onto the farm, as well as by disinfecting vehicles, clothing, tools, etc. that are brought onto the farm, these risks can be very effectively controlled.
然而,对于反刍动物而言,气溶胶传播也构成重大风险,而且这种传播方式更难控制。让我们来阐明气溶胶传播和飞沫传播的区别。
飞沫传播:直径大于5微米的颗粒,短距离(<1米)传播至易感动物。
气溶胶传播:直径小于5微米的颗粒,传播距离可达数百公里。
However, for ruminants, there is significant risk from aerosol transmission as well, something that is much more difficult to control. Let us clarify the difference between aerosol and droplet transmission.
Droplet transmission: particles >5 micrometers diameter, travelling short distances (<1 meter) to a susceptible animal.
Aerosol transmission: particles <5 micrometers diameter, travelling up to hundreds of km.
气溶胶传播的最大风险是来自猪传播到牛。这主要有两个原因:
猪呼出的病毒数量是牛羊的100-1000倍以上(Donaldson et al., 1970)。
牛羊吸入病毒后只需极少量即可感染(Donaldson et al., 2001),而且牛的呼吸量远大于猪羊。
The largest risk of aerosol transmission is from pigs to cattle. This is because of two factors:
Pigs shed 100-1000+ times more virus in their breath than cattle and sheep (Donaldson et al., 1970).
Cattle and sheep require a much smaller dose to be infected by inhalation than pigs (Donaldson et al., 2001), and cattle breathe a much higher volume of air than pigs or sheep.
表格 Table 1:感染口蹄疫病毒的猪、羊或小牛24小时内释放的空气中口蹄疫病毒峰值量。
Peak amount of airborne FMDV emitted in 24 hours by an infected pig, sheep, or heifer.
(表格改编自Table adapted from Donaldson & Alexandersen, 2002.)
物种 | 病毒株 Virus Strain | 数量 (TCID50 对数 / 24 小时) Amount (log TCID50 / 24 h) | 数量 (TCID50 / 24 小时) Amount (TCID50 / 24 h) |
猪Pig (90-100 kg) | C Noville | 8.6 | 400,000,000 |
猪 Pig (90-100 kg) | O UK 2001 | 6.1 | 1,250,000 |
小牛 Heifer (200 kg) | O UK 2001 | 4.3 | 20,000 |
羊 Sheep (30-40 kg) | OUK 2001 | 4.3 | 20,000 |
表格 Table 2: 24小时暴露期内感染不同动物所需的空气传播口蹄疫病毒的最低剂量。
Minimum doses of airborne foot and mouth disease virus required to infect different species during a 24-hour exposure period.
(表格改编自Table adapted from Donaldson & Alexandersen, 2002.)
动物 | 最小剂量 | 吸入速度 | 感染阈值浓度 |
牛Cow | 10 | 150 | 0.07 |
羊 Sheep | 10 | 15 | 0.7 |
猪 Pig | >800 | 50 | >16 |
牛和猪在出现临床症状时病毒排出量最大,而绵羊则往往在感染早期、临床症状出现之前就排出大量病毒(Donaldson et al., 1970)。
Cattle and pigs shed the largest amounts of virus when they have clinical signs, while sheep tend to shed the most virus early in the infection, before clinical signs appear (Donaldson et al., 1970).
影响气溶胶传播的因素
Factors Affecting Aerosol Transmission
某些血清型和毒株似乎比其它血清型和毒株产生气溶胶传播的风险更大。例如,Donaldson 1970 的研究表明,O 型和 C 型血清型的病毒排出量高于 A 型。此外,一项研究显示 亚洲1 型的气溶胶传播风险较低(Colenutt et al., 2016)。
Some serotypes and strains appear to have a greater risk of aerosol transmission than others. For instance, in Donaldson 1970, serotypes O and C were shed in larger amounts than type A. Additionally, Asia1 showed a lower risk for aerosol transmission in one study (Colenutt et al., 2016).
虽然目前尚无已发表的信息,但我看到一些报告指出,SAT-1 血清型比 O 型和 A 型血清型具有更高的气溶胶传播风险。
While we have no published information, I have seen reports that SAT-1 has a higher risk of aerosol transmission than serotypes O and A.
环境因素会影响病毒在空气中的存活时间。研究表明,较高的湿度(相对湿度高于 55%)、阴天无降水以及较低的气温有利于病毒在空气中存活更长时间,并增加气溶胶传播(Brown et al., 2022; Hugh-Jones & Wright, 1970)。此外,稳定的风速会使病毒聚集在气流中,导致下风处病毒浓度升高(Donaldson et al., 2001; Hugh-Jones & Wright, 1970)。
Environmental factors influence the survival of virus in the air. Higher humidity (>55% relative humidity), cloudy weather without precipitation, and cooler temperatures have been shown to favor longer survival of virus in the air and increased aerosol transmission (Brown et al., 2022; Hugh-Jones & Wright, 1970). In addition, steady winds allow for the virus to be concentrated in a plume, leading to an increased concentration of virus downwind (Donaldson et al., 2001; Hugh-Jones & Wright, 1970).
气溶胶传播风险于评估
Estimated Risk of Aerosol Transmission
由于影响气溶胶传播的因素多种多样,因此很难量化特定牧场在特定时间的风险。然而已有研究报告了已经的传播案例,其他研究则利用模型量化了风险。
Because the factors affecting aerosol transmission are varied, it is difficult to quantify the risk for a particular farm at a particular time. However, various studies have reported known transmission, and other studies have used models to quantify the risk.
已知的气溶胶传播通常在10km 以内(Henderson, 1969; Sellers & Forman, 1973),但也有许多病例记录在60km以上,甚至高达150km(Hugh-Jones & Wright, 1970)。1974年和1981年,口蹄疫被记录从法国布列塔尼传播到英国,估计传播距离为300-500km(Brown et al., 2022; Donaldson et al., 1982)。这是跨越英吉利海峡的传播;水路传播似乎比陆路传播更远。
Known transmission by aerosol has typically been within approximately 10 km (Henderson, 1969; Sellers & Forman, 1973), but with numerous cases documented at >60 km, even up to 150 km (Hugh-Jones & Wright, 1970). In 1974 and 1981, FMD was documented to spread from Brittany, France to England, with the estimated distances travelled being 300-500 km (Brown et al., 2022; Donaldson et al., 1982). This was over the English Channel; spread over water appears to be able to reach further distances than over land.
大多数关于长距离气溶胶传播的研究都来自英国。这或许部分归因于英国对该病毒的特殊研究兴趣,但英国的气候(凉爽多雨)也可能导致病毒更容易通过气溶胶传播,远超世界其它许多地区。
Most of the studies of aerosol transmission over long distances are from the UK. This may be in part due to a special research interest there, but it is also likely that the climate of the UK (cool with frequent rains) leads to greater spread by aerosol than many other parts of the world.
近年来,越来越多的研究利用口蹄疫病毒扩散的实验或模型来评估传播风险(Coffman et al., 2021; Colenutt et al., 2016; Donaldson et al., 2001; Donaldson & Alexandersen, 2002; Garner et al., 2006; Zhang et al., 2025)。这些研究普遍证实了以下观察结果:感染牧场周围 2 公里范围内风险最高,但在特定条下,10km 甚至 20km范围内的风险也不小(Coffman et al., 2021; Zhang et al., 2025)。
In recent years, there have been more studies using experiments or models of FMD dispersion to estimate risk of transmission (Coffman et al., 2021; Colenutt et al., 2016; Donaldson et al., 2001; Donaldson & Alexandersen, 2002; Garner et al., 2006; Zhang et al., 2025). These have generally confirmed the observations that the highest risk is within 2 km of an infected farm, though under the right conditions, the risk out to 10 or even 20 km is not small (Coffman et al., 2021; Zhang et al., 2025).
表3:美国堪萨斯州 10,000 头育肥场与源牧场距离不同处的感染风险估计
Table 3: Estimated Risk of Infection of 10,000 head Feedlot in Kansas at Varying Distances from the Source Farm
(表格改编自Table adapted from Coffman et al., 2021.)
距离 Distance | 1 km | 3km | 5km | 10km | 15km | 20km |
中位数概率 | 99.98% | 92.68% | 59.65% | 7.50% | 0.73% | 0.05% |
80% 置信区间 | 23-100% | 0-100% | 0-99% | 0-36% | 0-12% | 0-2% |
除了病毒血清型、病毒株和天气条件外,牧场上动物数量和密度的上升也会增加气溶胶传播的风险。对于排放病毒和下风处的牧场而言,情况都是如此。受感染牧场的动物数量越多,病毒传播量就越大,从而增加了下风处牧场的风险。此外,下风处牧场的动物数量越多,其中一只动物感染病毒的概率也就越高。
Apart from the virus serotype, virus strain, and weather conditions, an increase in the number and density of animals on a farm also increases the risk of aerosol transmission. This is true both for the farm emitting the virus, as well as the at-risk farm. More animals on the infected farm lead to higher quantities of virus being emitted, which increases the risk for downwind farms. Additionally, more animals on a downwind farm increases the probability that one of the animals will receive an infectious dose.
表2中估计的牛感染阈值浓度为 0.07 TCID50 / m3。2005 年的一项研究重新评估了 1967 年英国疫情的数据,并利用模型证实了最初的假设,即该疫情中许多牧场的感染是由气溶胶传播造成的((Gloster, Freshwater, et al., 2005);原始报告:(Hugh-Jones & Wright, 1970))。研究确定,大多数牧场确实是通过气溶胶传播感染的,但计算机模型预测大多数牧场的病毒浓度为 0.00005 - 0.0001 TCID50 / m3,远低于上述估计的阈值浓度。这意味着,即使模型预测的病毒浓度远低于感染所需的浓度,这些牧场仍然发生了感染。造成这种情况的原因尚不明确,但病毒有可能在小范围内聚集,从而增加感染风险(Gloster, Champion, et al., 2005)。将实际疫情与针对该疫情的模型预测进行比较表明,有很多变量我们无法充分了解,因此无法对风险进行非常精确的预测。
The estimated threshold concentration to infect cattle is 0.07 TCID50 / m3 in table 2. A 2005 study re-assessed data from the 1967 UK outbreak, using modelling to confirm the original hypothesis that many of the farms in that outbreak were due to aerosol transmission ((Gloster, Freshwater, et al., 2005); original report: (Hugh-Jones & Wright, 1970)). It was determined that most of the farms were truly infected by aerosol transmission, but the computer model predicted a virus concentration of 0.00005 - 0.0001 TCID50 / m3 at most of the farms, which is much lower than the threshold concentration estimated above. This means that, even though the model predicted a virus concentration much lower than what was required for infection, the farms were infected anyway. The reason for this is not certain, though it is possible that the virus could concentrate in small areas, leading to increased risk of infection (Gloster, Champion, et al., 2005). This comparison of an actual outbreak with modeled predictions for that outbreak demonstrates that there are many variables that cannot be known with adequate certainty to provide very accurate predictions of risk.
降低气溶胶传播风险
Reducing the Aerosol Risk
目前看来,似乎没有什么办法可以降低气溶胶传播的风险,尤其对牛而言。这是因为对进入牛羊畜棚内的空气进行过滤非常困难。猪舍更容易进行过滤,但这只能针对进入的空气进行过滤。因此,一个受感染的养猪场仍然会成为大量病毒的来源,进而感染其它牧场。
So far, there appears to be little that can be done to reduce the risk of aerosol transmission, especially for cattle. This is because it is difficult to filter the air coming into a cattle or sheep shed. Pig barns can be more easily filtered, though this is only done for incoming air. Thus, an infected pig farm would still be a source of large amounts of virus to infect other farms.
有一篇文章(Henderson, 1969)描述了一些牧场由于一片长着大树(约 15米高)的树林阻挡了来自被感染牧场的风,从而免受气溶胶传播的影响。然而,当风向改变时,这些牧场就会受到来自其他牧场的风的影响,因为它们没有来自那个方向的防风林。这表明,在牧场周围建造防风屏障可能具有一定的益处。例如,一些牧场已经具备冬季搭建防风屏障以抵御寒冷的能力。然而,这样做不太可能显著降低风险。
One of the articles (Henderson, 1969) described several farms being protected from aerosol transmission due to a section of wood with large trees (~15 m tall) blocking the wind from the infected farms. However, when the wind changed direction, these farms were infected by wind from other farms, as they did not have a windbreak from that direction. This indicates that there may be some benefit to constructing windbreaks along the perimeter of a farm. For instance, some farms already have the ability to put up windbreaks in the winter to protect from the cold. However, this is not likely to be able to noticeably reduce the risk.
根据同一篇文章,将动物圈养在舍内(禁止其外出)只能延缓牧场的感染,而无法阻止感染的发生。
According to the same article, keeping animals inside barns (not allowing outside access) only delayed, but did not prevent, infection of the farm.
减少牧场内气溶胶传播
Reducing Within-Farm Aerosol Spread
关于受感染牧场内空气传播的研究很少。此外,在牧场内建立有效的空气控制措施也并非易事。但根据我们已知的信息,如果牧场发生感染,将所有患病动物转移到下风处并尽可能与其它畜舍隔离的圈舍是合理的。这将有助于降低直接接触和气溶胶传播的风险。
There are few studies on airborne spread within infected farms. In addition, it is difficult to establish adequate controls of air within a farm. However, given what we do know, if a farm is infected, it is reasonable to move all sick animals to a pen that is downwind and separate from other barns as much as possible. This will help limit the transmission risk from direct contact as well as aerosol transmission.
亚临床携带者
Subclinical Carriers
牛病毒排出量最大的时期是在临床早期。但是,它们在出现临床症状前 1-3 天左右也可能开始排出病毒(Hayer et al., 2018)。相比之下,绵羊在出现临床症状前或接触病毒后36小时内排毒量更大,而且即使排毒,它们通常也没有临床症状(Donaldson et al., 1970)。此外,在英国的放牧条件下,绵羊似乎很少将口蹄疫传播到附近的牧场(Hugh-Jones & Wright, 1970)。这可能部分是因为附近牧场也饲养绵羊,因此疾病的检出率较低。
The period of shedding the most virus for cattle is early in the clinical stage. However, they can also shed virus for about 1-3 days before clinical signs (Hayer et al., 2018). In contrast, sheep shed virus more before clinical signs or within 36 hours of exposure, and often have no clinical signs even though they are shedding virus (Donaldson et al., 1970). In addition, it appears that in grazing conditions in England, sheep rarely transmitted FMD to neighboring farms (Hugh-Jones & Wright, 1970). This may be partly because the neighboring farms also had sheep, so detection of disease was low.
虽然临床患病动物排出的病毒量最大,但我们不能忽视亚临床感染的可能性。口蹄疫的亚临床感染有两种形式:新发感染和持续感染(Stenfeldt et al., 2020)。
新发感染是指动物排出病毒但没有临床症状。这种情况可能发生在疾病早期(潜伏期),也可能发生在接种过疫苗的动物(或对口蹄疫具有自然免疫力的动物)身上,这些动物从未出现临床症状,但也会排出病毒。
持续感染:已从临床疾病中康复的动物、接种疫苗后接触病毒的动物都可能发生持续感染。
持续感染的定义是:在初次感染后持续感染超过28天。
猪总是会4周内自然清除病毒,一般2周内即可。它们不会发生持续感染。
牛和羊可能发生持续感染。在持续感染中,病毒仅存在于鼻咽部。
While clinical animals shed the largest amounts of virus, we cannot ignore the possibility of subclinical infection. There are two forms of subclinical infection with FMD: neoteric infection, and persistent infection (Stenfeldt et al., 2020).
Neoteric infection refers to an animal that is shedding virus but has no clinical signs. This can be early in the course of disease (during the incubation period), or this can happen in vaccinated animals (or animals naturally immune to FMD) that never show clinical disease but shed the virus.
Persistent Infection: animals that have recovered from clinical disease, as well as vaccinated animals that have been exposed, can both be persistently infected.
Persistent infection is defined as infection for >28 days after the initial infection.
Pigs always naturally clear the virus within 4 weeks, and usually within 2 weeks. They cannot be persistently infected.
Cows and sheep can be persistently infected. In persistent infections, the virus is only found in the nasopharynx.
与牛病毒性腹泻(BVD)不同,持续感染的动物不会排出足够数量的病毒来感染其他动物。许多实验都试图证明持续感染的动物可以感染未感染的动物,但这些实验均未成功。然而,这些动物携带的是活病毒:如果从持续感染的动物中分离出病毒,就可以用它人工接种未感染的动物,从而引发疾病。但是,我们目前尚无证据表明持续感染的动物能够自然传播该疾病(Stenfeldt et al., 2020)。
In contrast to BVD, persistently-infected animals do not shed the virus in sufficient quantities to infect other animals. Numerous experiments have attempted to show that persistently-infected animals can infect naïve animals, but none of them have succeeded. However, these animals are carrying live virus: if the virus is isolated from a persistently-infected animal, it can be used to artificially inoculate naïve animals and cause disease. However, we have no proof that persistently infected animals can transmit the disease naturally (Stenfeldt et al., 2020).
因此,亚临床感染的主要关注点在于新发感染:即在疾病早期、临床症状出现之前就排出病毒,或者接种过疫苗的动物即使没有出现临床症状,也仍然会排出病毒。
Thus, the primary concern with subclinical infections are the neoteric infections: either shedding virus early in the disease process before clinical signs, or vaccinated animals that have been exposed and are shedding virus, even without clinical signs.
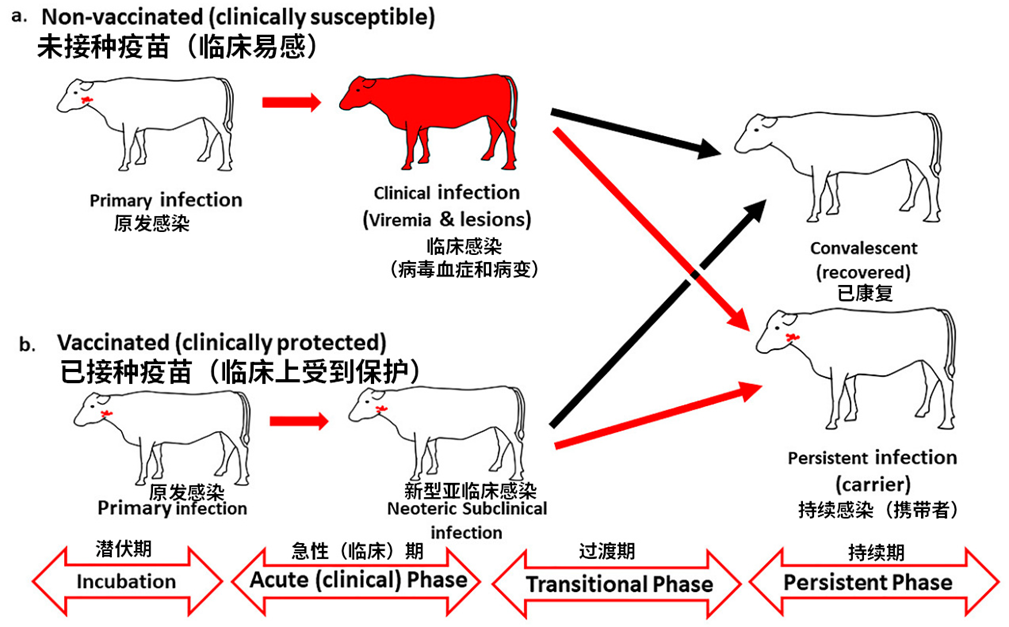
(改编自 (Stenfeldt et al., 2020)。 Adapted from (Stenfeldt et al., 2020).)
接种疫苗 Vaccination
疫苗接种通常是由灭活病毒颗粒与佐剂混合。这些疫苗能有效预防临床疾病,但不能完全预防亚临床感染(Stenfeldt et al., 2020)。此外,与所有疫苗一样,它们并不能100%预防临床疾病:一小部分接种疫苗的动物仍可能出现临床疾病。
Vaccination typically uses inactivated virus particles mixed with an adjuvant. These vaccines are effective at preventing clinical disease, but are not able to completely prevent subclinical infection (Stenfeldt et al., 2020). In addition, as with all vaccines, they do not prevent 100% of clinical disease: a small proportion of vaccinated animals may still get clinical disease.
(Hayer et al., 2018)记录了印度一个奶牛场中口蹄疫的动态,该奶牛场已接种了针对疫情毒株的疫苗。换句话说,尽管疫苗毒株(O/IND/R2/75)目的在于预防疫情毒株(O/ME-SA/Ind-2001d),但该牧场仍然爆发了疫情。在1836头定期接种疫苗的成年奶牛中,有222头(12%)在39天内出现临床症状。在该牛群中,疫苗的有效率为88%,但仍然发生了疫情。该毒株的R0估计值为67。也就是说,在未感染的牛群中,一例病牛会传染给其它67头牛。然而,在接种疫苗的牛群中,R*估计值为8-10。这意味着即使牛群已接种疫苗,一头牛仍可能传染给其它8-10头牛。
(Hayer et al., 2018) documented the dynamics of FMD in a dairy herd in India that was vaccinated for the outbreak strain. In other words, even though the vaccine strain (O/IND/R2/75) was designed to protect against the outbreak strain (O/ME-SA/Ind-2001d), the farm still experienced an outbreak. Of 1836 regularly-vaccinated adult dairy cows, 222 (12%) had clinical signs over a 39-day period. In this herd, the vaccine was 88% effective, yet they still had an outbreak. The R0 for this strain was estimated to be 67. That is, in a naïve population, one case would infect 67 other animals. However, in this vaccinated population, the R* was estimated to be 8-10. This means that even though the herd was vaccinated, one animal could infect 8-10 other animals.
值得注意的是,感染风险最高的奶牛是未怀孕的奶牛。亚临床传染期与临床传染期一样长,这表明很多传播是在没有临床症状的情况下发生的。
It is worth noting that the cows at highest risk of infection were non-pregnant cows. It is also worth noting that the subclinical infectious period was as long as the clinical infectious period, indicating that much transmission was occurring without clinical signs.
这项研究表明,疫苗接种对预防口蹄疫有很大帮助,但不能将其作为唯一的控制手段。
This study shows us that vaccination is a big help to preventing FMD, but it cannot be relied upon as the sole method of control.
提高疫苗效力
Improving Vaccine Efficacy
尽管没有任何疫苗接种方案能达到100%的有效性,但我们可以通过设计合适的疫苗接种方案来提高有效免疫的可能性。多种因素会影响个体动物接种疫苗后能否成功产生保护性免疫反应。
Even though no vaccination program will be 100% effective, we can improve the likelihood of effective immunization by designing appropriate vaccination protocols. Various factors affect whether an individual animal successfully mounts a protective response when vaccinated.
动物的整体健康状况会影响免疫反应。虽然各类的研究有限,但以下因素通常被认为会降低疫苗的免疫反应:
营养不良或能量负平衡。瘦弱或者体重下降的动物不太可能产生有效的免疫反应。除了足够的蛋白质和能量外,足够水平的维生素和矿物质也是很有必要的。
其他疾病:任何其他疾病(尤其是传染病)都会降低疫苗的免疫反应。对于导致免疫抑制的疾病,例如病毒性腹泻(BVD)和牛白血病病毒(BLV),尤其如此。副结核和肺结核等慢性疾病也可能降低免疫反应。
应激事件,例如断奶、产犊、运输,甚至转群。
免疫抑制药物,特别是糖皮质激素(氢化可的松、地塞米松等)。
年龄:免疫反应会随着年龄的增长而下降。
The overall health status of the animal affects the vaccine response. Though specific studies are limited, these factors are generally considered to decrease the immune response to a vaccine:
Poor nutrition or negative energy balance. Animals that are thin or in a state of weight loss are less likely to mount an effective immune response. Besides energy and protein, adequate levels of vitamins and minerals are necessary.
Other disease: any other disease (especially infectious disease) can reduce the immune response to a vaccine. This is especially true for diseases that cause immune suppression such as BVD, BHV, and BLV. Chronic diseases such as Johne's and TB would also likely reduce the immune response.
Stressful events such as weaning, calving, transportation, or even pen moves.
Immune-suppressive drugs, especially glucocorticoids (hydrocortisone, dexamethasone, etc.).
Age: immune response declines with age.
有些因素是无法控制的,但是我们能做的就是改善营养状况,减少其他疾病负担,并降低应激有所帮助。
Some of these factors are uncontrollable. However, what we can do to improve nutritional status, reduce the burden of other diseases, and reduce stress will help.
母牛安抚物质 (MBAS) 可降低应激反应,已被证明能增强疫苗接种后的免疫反应。美国一项关于馡安美® (FerAppease®) 的研究((Pickett et al., 2024),个人交流)表明,在接种疫苗时使用馡安美可显著提高牛对曼氏杆菌类毒素、梭菌类毒素和 IBR/PI3 滴鼻疫苗的抗体滴度。美国牧场正在使用馡安美来延长疫苗的有效时间,从而减少接种频率。
Maternal bovine appeasing substance (MBAS), which reduces the negative responses to stress, has been shown to improve the immune response to vaccination. In US research on FerAppease® ((Pickett et al., 2024), personal communication), the use of FerAppease at the time of vaccination greatly increased antibody titers to Mannheimia toxoid, Clostrisdium toxoid, and IBR / PI3 intranasal vaccines. Farms in the US are using FerAppease to extend the duration of efficacy of vaccines, allowing them to vaccinate less frequently.
血清型和拓扑型
Serotypes & Topotypes
众所周知,针对A型和O型血清型的疫苗不能预防SAT-1型血清型。这是因为灭活病毒疫苗通常引起针对表面蛋白的免疫反应,而不同血清型的表面蛋白(SP)并不相同。即使同一血清型,不同拓扑型的病毒之间也存在不同程度的交叉保护。
As we know, vaccines for serotypes A and O do not protect against serotype SAT-1. This is because inactivated virus vaccines typically induce an immune response to the surface proteins, and the surface proteins (SP) are not the same across serotypes. Even within serotypes, different topotypes of virus have varying degrees of cross-protection.
2025 年末,SAT-1 拓扑型 I 和拓扑型 III 均在伊朗和土耳其传播(FAO, 2026)。这意味着针对一种拓扑型的疫苗可能无法充分预防另一种SAT-1 拓扑型。目前我还不清楚中国内新生产的灭活疫苗包含哪种拓扑型。
SAT-1 topotype I and topotype III are both circulating in Iran and Turkey in late 2025 (FAO, 2026). This means that it is possible that the vaccine for one topotype will not adequately protect against the other topotype of SAT-1. I do not yet know which topotype is included in the new inactivated vaccine produced in China.
新型疫苗技术
New Vaccine Technology
据报道,一种新型亚单位疫苗(Mu et al., 2026)在国内首次使用。这种亚单位疫苗与T细胞表位结合,能有效刺激细胞介导的免疫反应。
According to reports, a new type of sub-unit vaccine is being used for the first time in China (Mu et al., 2026). This sub-unit vaccine is combined with T cell epitopes that effectively stimulate cell-mediated immunity.
作为背景,获得性免疫系统主要由两大部分组成:体液免疫(由B细胞产生抗体,并受Th2辅助细胞刺激)和细胞介导免疫(由Th1辅助细胞刺激的杀伤T细胞)。细胞介导免疫用于清除细胞内病原体(例如病毒),而体液免疫(抗体)主要用于对抗细胞外病原体(例如细菌)。
As background, there are 2 main arms of the acquired immune system: humoral (antibodies produced by B cells, stimulated by Th2 helper cells) and cell-mediated immunity (killer T cells stimulated by Th1 helper cells). Cell-mediated immunity is used to clear intra-cellular pathogens (such as viruses), whereas the humoral side (antibody) side of the immune system is used primarily for extracellular pathogens (such as bacteria).
灭活疫苗主要刺激体液免疫(抗体),因为灭活病毒不会进入细胞,而是停留在细胞外。抗体在血清中循环,有助于中和病毒,但其预防感染的效果不那么理想。
Inactivated vaccines mostly stimulate humoral immunity (antibodies), because the inactivated virus does not enter the cells: it stays outside the cells. Antibodies produced circulate in the serum and can help neutralize virus, but they are not as effective at preventing infection.
但是根据这项(Mu et al., 2026)初步研究,这种亚单位疫苗似乎具有两个优势:
所用亚单位来源于口蹄疫病毒的非结构蛋白(NSP),这些蛋白高度保守(在不同血清型和毒株中相同)。这使得该亚单位疫苗能够预防多种血清型。该研究仅测试了A型和O型血清型,但它很可能也能预防SAT-1型血清型。
刺激细胞介导的免疫反应,这可能比传统疫苗提供更强的保护作用。有研究表明,完全清除病毒的牛比持续感染的牛具有更强的Th1反应(Stenfeldt et al., 2020)。因此,能更好地刺激细胞介导免疫的疫苗可能在预防持续性感染方面更有效。
However, based on this initial study (Mu et al., 2026), this subunit vaccine appears to have 2 advantages:
The subunits used are from the non-structural proteins (NSP) in FMDV, which are highly conserved (are the same across serotypes and strains). This allows the subunit vaccine to protect against multiple serotypes. Only serotypes A and O were tested in the study, but it is likely to protect against SAT-1 as well.
Stimulation of the cell-mediated immune response, which may provide increased protection compared to traditional vaccines. Th1 responses have been shown to be stronger in animals that completely clear the virus than in animals that become persistently infected (Stenfeldt et al., 2020). Therefore, it may be that vaccines that better stimulate cell mediated immunity will be more effective at protecting against persistent infections.
该研究仅报告了猪的实验结果。因此,我们尚不清楚它对牛是否也有效。此外,该研究仅在实验室进行。田间试验将验证该疫苗对多种动物(猪、牛、羊、山羊)的有效性和安全性。
The study only reports experiments in pigs. Therefore, we have yet to see if it is effective in cattle as well. Additionally, the study was only conducted in a laboratory. Field trials will show whether the vaccine is both effective and safe across a range of species (pigs, cattle, sheep, goats).
如果该疫苗被证实安全有效,那么有望通过它提供广谱保护,甚至可能减少亚临床(新发)感染,从而显著控制口蹄疫。
If this vaccine proves to be safe and effective, it may significantly help control FMD by providing broad-spectrum protection, and possibly even reducing subclinical (neoteric) infections.
皮内疫苗接种
Intradermal Vaccine Delivery
面对新型口蹄疫毒株的爆发,一个常见的问题是疫苗供应不足,无法及时控制疫情。为了减少疫苗需求量,选择性地为高危群体接种疫苗以及采用低剂量皮内接种是潜在的解决方案。
In the face of an outbreak with a new FMD strain, a common problem is the lack of adequate vaccine soon enough to contain the outbreak. In order to reduce the amount of vaccine needed, selective vaccination of high-risk groups and reduced-dose intradermal vaccination are potential options.
大量研究(Eblé et al., 2009; Hwang et al., 2019; Kim et al., 2026; Le Luduec et al., 2016; Pandya et al., 2012)表明,低剂量皮内接种疫苗可以有效预防口蹄疫。据我所知,这种方法还未在田间实施。皮内接种需要特殊的设备,例如使用压缩二氧化碳注射疫苗的无针疫苗系统。另一种方法(尚未进行研究)是使用麦克林托克注射器。这是一种专为皮内注射结核因素而设计的多剂量注射器。
Numerous studies (Eblé et al., 2009; Hwang et al., 2019; Kim et al., 2026; Le Luduec et al., 2016; Pandya et al., 2012) have shown that vaccinating with a reduced dose intradermally can achieve good protection against FMD. As far as I know, this has not been done in the field. Intradermal vaccination requires special equipment such as needle-less vaccine systems that use compressed CO2 to inject the vaccine. An alternative (not yet studied) could be to use McClintock syringes. These are multiple-dose syringes designed for intradermal injection of tuberculin.
畜群内感染管理
Within-Herd Management of Infection
目前针对畜群感染后的管理方案研究较少。其他一些研究建议隔离感染动物并进行消毒,因此本文仅讨论针对已感染牧场的治疗和控制的新思路。
Little research has been done to address management options once a herd is infected. There are suggestions elsewhere with details about segregation of infected animals and disinfection, so this discussion is limited to new ideas regarding treatment and control on an infected farm.
临床病例治疗
Treatment of Clinical Cases
基于其他国家的报告,典型的病例治疗方法包括:
提供柔软的饲料(牧草、苜蓿青贮、青贮饲料等)和充足的饮水。
使用抗炎药(氟尼辛、卡洛芬、美洛昔康等)控制疼痛。但是,如果止痛导致过度行走,可能会对蹄部造成更大的损伤。
使用温和的消毒剂(稀释的高锰酸钾)清洁损伤部位。
使用抗生素(例如土霉素)治疗重症病例(伴有蹄部损伤或细菌感染)。
Based on reports from other countries, typical treatment of cases includes:
Provide soft feed (grass, haylage, silage, etc.) and adequate water
Anti-inflammatories (flunixin, carprofen, meloxicam, etc.) for pain control. However, if pain control leads to excessive walking, it may cause more damage to hooves.
Clean lesions with mild antiseptics (dilute potassium permanganate).
Treat severe cases (with feet lesions or with evidence of bacterial infection) with antibiotics such as oxytetracycline.
为了加快康复,可以在口腔损伤处使用小苏打(碳酸氢钠)、蜂蜜和小米粉。碳酸氢钠的高pH值可以灭活病毒颗粒,而蜂蜜和指粟(Eleusine coracana)粉则能促进病变更快愈合。病例报告(Gakuya et al., 2010)表明,奶牛在3天内即可恢复采食。另有报道称,用醋或柠檬酸漱口(Lubroth, 2002)也有助于加快愈合,其原理同样是通过灭活口腔中的病毒颗粒。
In order to speed recovery, baking soda (sodium bicarbonate), honey, and finger millet (Eleusine coracana) flour can be used on oral lesions. The sodium bicarbonate inactivates virus particles by the high pH, while honey and millet flour encourage faster healing of lesions. Case reports (Gakuya et al., 2010) indicate that cows return to eating within 3 days. Oral rinsing with vinegar or citric acid (Lubroth, 2002) has also been reported to help speed healing, again by inactivating virus particles in the mouth.
这些治疗方法的局限性之一是需要每天至少对每头感染动物进行一次处理。如果同时感染的动物数量较多,则可能无法做到这一点。
One of the limitations of these treatment methods is the need to handle each infected animal at least once daily. This may become impossible if many animals are infected at once.
治疗的主要目标之一是帮助患病动物尽快恢复进食。它们拒食的主要原因是口腔疼痛。因此,喂食软饲料并使用消炎药来减轻疼痛会有所帮助。
One of the main goals of treatment is to help sick animals return to eating as soon as possible. The main reason they refuse to eat is because of the pain in their mouth. Thus, feeding soft feeds and using anti-inflammatories to reduce pain can help.
另一个导致干物质采食量减少的常见原因是应激。疼痛和应激的影响往往同时出现,因此很难区分二者的影响。然而,使用母牛专用镇静剂(例如馡安美)来降低应激感知,可能有助于增加采食量,从而减轻疾病的严重程度。这种现象已在难产后的母牛和患有肺炎的犊牛身上观察到。由于其药效持续时间为 14 天,因此每个病例只需使用一次即可。
Another well-known cause of reduced dry matter intake is stress. It is difficult to separate the effect of pain and the effect of stress because they often go together. However, reducing the perception of stress through the use of maternal bovine appeasing substance (such as FerAppease) may also be useful for increasing feed intake, leading to reduced severity of disease. This phenomenon has been noted when used with heifers after dystocia and on calves with pneumonia. Because the duration of efficacy is 14 days, it would only need to be applied once per case.
饮用水酸化
Acidification of Drinking Water
基于以上的信息,我建议将牧场上的饮用水酸化至弱酸性(PH值约为5.8-6.0)或弱碱性(PH值约为9)。这可能有助于降低严重程度以及牧场上口蹄疫的传播。这可以通过在水中添加柠檬酸、醋酸(醋)或小苏打轻松实现。所需的酸或碱的量将取决于水中其他溶解矿物质的含量,这些矿物质起到缓冲作用。
Based on the above information, I propose that making the drinking water on farms slightly acidic (pH ~5.8-6.0) or slightly basic (pH ~9) may help to reduce the severity and spread of FMD on farms. This could be easily done by adding citric acid, acetic acid (vinegar), or baking soda to the water. The amount of acid or base needed would be influenced by the amount of other dissolved minerals in the water which would act as a buffer.
稍微酸化水的好处是,这样可以对更多牛产生效果,而无需单独治疗每头牛。
The advantage of slightly acidifying the water is that this will have an effect for many more animals without the need to individually treat every animal.
我认为酸化/碱化饮用水将会有两方面益处:
酸化/碱化水可以减少牛口腔中的病毒载量。无论牛是接触过其它牛的病毒颗粒,还是已经临床感染,这都能降低口腔中的病毒载量,从而减轻疾病的严重程度(如上文所述)。
降低通过水槽将病毒传播给其他牛的风险。被感染的动物会在唾液中排出大量病毒,导致包括水槽在内的环境受到污染。酸化/碱化饮用水可以降低水槽中的病毒载量,从而可能降低传播风险。
I believe that acidifying / alkalinizing the drinking water may be beneficial in two ways:
The acidic / alkaline water would reduce the viral load in the mouth of cattle. Whether they exposed to virus particles from other cows or are clinically infected, this would reduce the viral load in the mouth, leading to reduced severity of disease (as described above for treatments).
The risk of spreading virus to other cattle via the water trough would be reduced. Infected animals shed large amounts of virus in their saliva, leading to contamination of their environment, including water troughs. Acidification / alkalization of the drinking water would reduce the viral load in the water trough, potentially reducing the risk of transmission.
饲喂柠檬酸、醋酸和碳酸氢钠对反刍动物是安全的。
柠檬酸在瘤胃里迅速代谢为醋酸(Wright, 1971),醋酸作为能量来源通过瘤胃壁被吸收。反刍动物试验(Chandrika et al., 2022; Shen et al., 2019)表明,饲喂柠檬酸可改善动物的瘤胃功能。因此,我认为使用柠檬酸酸化饮用水的安全性无需担忧。
常规饲喂碳酸氢钠的量为每天100g以上,并可在食槽中自由采食
Feeding citric acid, acetic acid, and sodium bicarbonate is safe for ruminants.
Citric acid is metabolized quickly to acetic acid in the rumen (Wright, 1971), which is absorbed as a source of energy across the rumen wall. Trials in ruminants (Chandrika et al., 2022; Shen et al., 2019) have shown improved rumen function of animals fed citric acid. Therefore, I have no reason to be concerned about the safety of using citric acid to acidify drinking water.
Sodium bicarbonate is routinely fed in amounts of 100+ grams / day, as well as offered free-choice in the feed bunk.
酸通常比碱对伤口(例如口腔溃疡)的刺激性更大,但pH值为6应该不会产生刺激。根据对SAT-1血清型pH稳定性的分析(Scott et al., 2019),pH值为6.1会导致大多数SAT-1血清型完全失活,而pH值为9.1则影响不大。因此,我认为酸化(pH值约为5.8-6.0)比碱化(pH值约为9)更有效。
Acids are generally more irritating to wounds (such as oral ulcers) than bases, but a pH of 6 should not be irritating. Based on analysis of the pH stability of SAT serotypes (Scott et al., 2019), a pH of 6.1 causes complete deactivation of most SAT-1 serotypes, whereas a pH of 9.1 does not have much effect. Therefore, I believe that acidification (pH ~6) would be more effective than alkalization (pH~9).
总结
Summary
虽然限制牲畜流动、消毒和接种疫苗是控制口蹄疫的基础,但深入了解口蹄疫的细节以及潜在防控措施有助于提高防控策略的有效性。本文介绍了有关口蹄疫传播(特别是气溶胶传播)、亚临床感染、疫苗接种和牧场感染管理等方面更多信息。基于这些信息,我们提出了几种改进控制口蹄疫的潜在方案,包括:
在牧场周围(特别是靠近疫情爆发区的牧场)设置防风屏障,以降低气溶胶传播的风险。
使用皮内注射疫苗,以使疫苗供应不足的情况下可以为更多的动物接种。
在接种疫苗时使用母牛安抚信息素,以增强免疫反应,并提高临床病牛的采食量。
在饮用水中添加柠檬酸,将pH值降至5.8-6.0,以减少水源污染并减轻临床病牛的严重程度。
While movement restrictions, disinfection, and vaccination form the basis of controlling FMD, having a deeper understanding of the details of FMD and potential control methods can help to increase the effectiveness of control strategies. In this article, we have presented additional information regarding transmission (especially aerosol), subclinical infection, vaccination, and management of infected farms. Based on the additional information, several potential options for improving FMD control were presented, including:
Use of wind barriers around farms (especially those close to outbreak areas) to decrease the risk of aerosol transmission.
Use of intradermal vaccination in order to vaccinate more animals with limited doses of vaccine.
Use of maternal bovine appeasing substance at vaccination to improve immune response, and for clinical cases to increase feed intake.
Use of citric acid in the drinking water to drop the pH to 6, in order to reduce contamination of the water source and to reduce severity of clinical cases.
参考文献 References
Arzt, J., Sanderson, M. W., & Stenfeldt, C. (2024). Foot-and-Mouth Disease. Veterinary Clinics of North America - Food Animal Practice, 40(2), 191–203. https://doi.org/10.1016/J.CVFA.2024.01.001/ASSET/A817F6B1-A4C5-4E51-9EC5-39337FD8C7AA/MAIN.ASSETS/GR1.SML
Brown, E., Nelson, N., Gubbins, S., & Colenutt, C. (2022). Airborne Transmission of Foot-and-Mouth Disease Virus: A Review of Past and Present Perspectives. Viruses, 14(5), 1009. https://doi.org/10.3390/V14051009
Chandrika, B., Suryanarayna, M., Devasena, B., & Reddy, Y. R. (2022). Effect of citric acid supplementation on rumen fermentation pattern, milk composition and blood biochemical profile in cross bred cattle. The Pharma Innovation Journal, (3), 427–431.
Coffman, M. S., Sanderson, M. W., Dodd, C. C., Arzt, J., & Renter, D. G. (2021). Estimation of foot-and-mouth disease windborne transmission risk from USA beef feedlots. Preventive Veterinary Medicine, 195, 105453. https://doi.org/10.1016/J.PREVETMED.2021.105453
Colenutt, C., Gonzales, J. L., Paton, D. J., Gloster, J., Nelson, N., & Sanders, C. (2016). Aerosol transmission of foot-and-mouth disease virus Asia-1 under experimental conditions. Veterinary Microbiology, 189, 39–45. https://doi.org/10.1016/J.VETMIC.2016.04.024
Donaldson, A. I., & Alexandersen, S. (2002). Predicting the spread of foot and mouth disease by airborne virus. OIE Revue Scientifique et Technique, 21(3), 569–575. https://doi.org/10.20506/RST.21.3.1362
Donaldson, A. I., Alexandersen, S., Sørensen, J. H., & Mikkelsen, T. (2001). Relative risks of the uncontrollable (airborne) spread of FMD by different species. Veterinary Record, 148(19), 602–604. https://doi.org/10.1136/VR.148.19.602
Donaldson, A. I., Gloster, J., Harvey, L. D., & Deans, D. H. (1982). Use of prediction models to forecast and analyse airborne spread during the foot-and-mouth disease outbreaks in Brittany, Jersey and the Isle of Wight in 1981. The Veterinary Record, 110(3), 53–57. https://doi.org/10.1136/VR.110.3.53
Donaldson, A. I., Herniman, K. A. J., Parker, J., & Sellers, R. F. (1970). Further investigations on the airborne excretion of foot-and-mouth disease virus. The Journal of Hygiene, 68(4), 557–564. https://doi.org/10.1017/S0022172400042480
Eblé, P. L., Weerdmeester, K., van Hemert-Kluitenberg, F., & Dekker, A. (2009). Intradermal vaccination of pigs against FMD with 1/10 dose results in comparable vaccine efficacy as intramuscular vaccination with a full dose. Vaccine, 27(8), 1272–1278. https://doi.org/10.1016/j.vaccine.2008.12.011
FAO. (2026). FAO alerts countries in Asia and the Pacific to enhance preparedness for foot-and-mouth disease SAT1.
Gakuya, D. W., Mulei, C. M., & Wekesa, S. B. (2010). Use of Ethnoveterinary Remedies in the Management of Foot and Mouth Disease Lesions in a Diary Herd. African Journal of Traditional, Complementary, and Alternative Medicines, 8(2), 165. https://doi.org/10.4314/AJTCAM.V8I2.63204
Garner, M. G., Hess, G. D., & Yang, X. (2006). An integrated modelling approach to assess the risk of wind-borne spread of foot-and-mouth disease virus from infected premises. Environmental Modeling and Assessment, 11(3), 195–207. https://doi.org/10.1007/S10666-005-9023-5/METRICS
Gloster, J., Champion, H. J., Mansley, L. M., Romero, P., Brough, T., & Ramirez, A. (2005). The 2001 epidemic of foot-and-mouth disease in the United Kingdom: epidemiological and meteorological case studies. The Veterinary Record, 156(25), 793–803. https://doi.org/10.1136/VR.156.25.793
Gloster, J., Freshwater, A., Sellers, R. F., & Alexandersen, S. (2005). Re-assessing the likelihood of airborne spread of foot-and-mouth disease at the start of the 1967-1968 UK foot-and-mouth disease epidemic. Epidemiology and Infection, 133(5), 767. https://doi.org/10.1017/S0950268805004073
Hayer, S. S., VanderWaal, K., Ranjan, R., Biswal, J. K., Subramaniam, S., Mohapatra, J. K., Sharma, G. K., Rout, M., Dash, B. B., Das, B., Prusty, B. R., Sharma, A. K., Stenfeldt, C., Perez, A., Delgado, A. H., Sharma, M. K., Rodriguez, L. L., Pattnaik, B., & Arzt, J. (2018). Foot-and-mouth disease virus transmission dynamics and persistence in a herd of vaccinated dairy cattle in India. Transboundary and Emerging Diseases, 65(2), e404–e415. https://doi.org/10.1111/TBED.12774
Henderson, R. J. (1969). The outbreak of foot-and-mouth disease in Worcestershire. An epidemiological study: with special reference to spread of the disease by wind-carriage of the virus. The Journal of Hygiene, 67(1), 21–33. https://doi.org/10.1017/S0022172400041383
Hugh-Jones, M. E., & Wright, P. B. (1970). Studies on the 1967-8 foot-and-mouth disease epidemic. The relation of weather to the spread of disease. The Journal of Hygiene, 68(2), 253–271. https://doi.org/10.1017/S0022172400028722
Hwang, J. H., Lee, K. N., Kim, S. M., Lee, G., Moon, Y., Kim, B., Lee, J. S., & Park, J. H. (2019). Needleless intradermal vaccination for foot-and-mouth disease induced granuloma-free effective protection in pigs. Journal of Veterinary Science, 20(3), e29. https://doi.org/10.4142/JVS.2019.20.E29
Kim, D. W., Lee, S. Y., Kim, T. J., Kim, H., Hwang, J. H., Park, S. Y., Ko, Y. J., Lee, Y. H., Park, J. H., & Park, S. H. (2026). Evaluation of an Effective Intradermal Foot-and-Mouth Disease Vaccine for Early Protection. Vaccines, 14(3). https://doi.org/10.3390/VACCINES14030263
Le Luduec, J.-B., Debeer, S., Piras, F., Andréoni, C., Boudet, F., Laurent, P., Kaiserlian, D., & Dubois, B. (2016). Intradermal vaccination with un-adjuvanted sub-unit vaccines triggers skin innate immunity and confers protective respiratory immunity in domestic swine. Vaccine, 34(7), 914–922. https://doi.org/10.1016/j.vaccine.2015.12.058
Lubroth, J. (2002). Foot-and-mouth disease. A review for thepractitioner. The Veterinary Clinics of North America. Food Animal Practice,18(3), 475–499, viii. https://doi.org/10.1016/s0749-0720(02)00036-1
Mu, S., Shang, S., Dong, H., Wang, S., Chen, L., Zhang, Z., Bai, M., Teng, Z., Ding, Y., Zhang, Y., Guo, H., & Sun, S. (2026). T cell responses to nonstructural proteins promote cross-serotype immunity to foot-and-mouth disease virus. MBio. https://doi.org/10.1128/MBIO.03586-25
Pandya, M., Pacheco, J. M., Bishop, E., Kenney, M., Milward, F., Doel, T., & Golde, W. T. (2012). An alternate delivery system improves vaccine performance against foot-and-mouth disease virus (FMDV). Vaccine, 30(20), 3106–3111. https://doi.org/10.1016/j.vaccine.2012.02.049
Pickett, A. T., Cooke, R. F., de Souza, I. S., & Mackey, S. J. (2024). Administering the maternal bovine appeasing substance improves overall productivity and health in high-risk cattle during a 60-d feedlot receiving period. Journal of Animal Science, 102. https://doi.org/10.1093/JAS/SKAE221
Scott, K. A., Maake, L., Botha, E., Theron, J., & Maree, F. F. (2019). Inherent biophysical stability of foot-and-mouth disease SAT1, SAT2 and SAT3 viruses. Virus Research, 264, 45–55. https://doi.org/10.1016/J.VIRUSRES.2019.02.012
Sellers, R. F., & Forman, A. J. (1973). The Hampshire epidemic of foot-and-mouth disease, 1967. The Journal of Hygiene, 71(1), 15–34. https://doi.org/10.1017/S0022172400046179
Sellers, R. F., Herniman, K. A., & Mann, J. A. (1971). Transfer of foot-and-mouth disease virus in the nose of man from infected to non-infected animals. The Veterinary Record, 89(16), 447–449. https://doi.org/10.1136/VR.89.16.447-A
Shen, Y. Z., Ding, L. Y., Chen, L. M., Xu, J. H., Zhao, R., Yang, W. Z., Wang, H. R., & Wang, M. Z. (2019). Feeding corn grain steeped in citric acid modulates rumen fermentation and inflammatory responses in dairy goats. Animal, 13(2), 301–308. https://doi.org/10.1017/S1751731118001064
Stenfeldt, C., Arzt, J., Stenfeldt@usda, C., Gov, C. S., Arzt@usda, J., & Gov, J. A. (2020). The Carrier Conundrum; A Review of Recent Advances and Persistent Gaps Regarding the Carrier State of Foot-and-Mouth Disease Virus. Pathogens 2020, Vol. 9, Page 167, 9(3), 167. https://doi.org/10.3390/PATHOGENS9030167
Wright, D. E. (1971). Citric Acid Metabolism in the Bovine Rumen. Applied Microbiology, 21(2), 165. https://doi.org/10.1128/AM.21.2.165-168.1971
Zhang, J., Wang, J. L., Jin, Z., Wei, Y. R., & Zhao, Z. Z. (2025). Quantifying aerosol transmission distance for foot-and-mouth disease virus. Advances in Continuous and Discrete Models, 2025(1), 176-. https://doi.org/10.1186/S13662-025-04035-2/FIGURES/15

公司地址

All Rights Reserved
陕西省西安市高新区唐延南路11号逸翠园i都会1幢1单元101025室
电邮:stephan@meimusheng.com

